 
Energy of activation = Threshold energy – Energy of reactantĬheck out the Formula Collection of various concepts from different subjects altogether at a reliable site. Contrary to potential energy, the kinetic energy of an object is relative to other stationary and moving objects present in its immediate environment. (1) The dimensional formula of mass M1L0T0. The dimensional formula of kinetic energy can be given by M1L2T-2 Where M mass L Length T Time It can be derived as follows Kinetic energy K.E. Rate of Reaction in the form of a Chemical reaction: The kinetic energy formula is given by K.E. Try to memorize the Simple Formulas on Chemical Kinetics provided as they form a stronger base during your study.ġ. After reading the text on Gas Temperature and the Kinetic Theory of Gases, we are now going to do some mathematics using the kinetic energy formula. 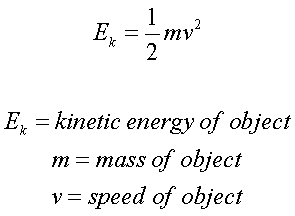
Take your preparation to the next level by employing the Chemical Kinetic Formulae over here and solve the reactions quickly. 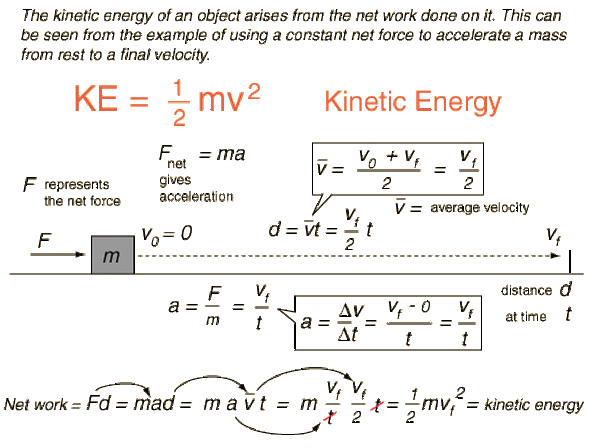
Going through the Chemical Kinetics Cheat Sheet you will have an overview of what Chemical Kinetics is exactly. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
